In the context of the healthcare industry growing strongly, job import medical equipment becoming an indispensable activity of many businesses to meet the increasing demand for equipment, new technology. However, to ensure import operations go smoothly, in accordance with the law, Enterprises need to carry out application procedures import license accurately and completely. Below are detailed instructions to help businesses understand the necessary steps to apply import license Medical equipment in 2025.
Article Content
- 0.1 Overview of medical equipment import trends in 2025
- 0.2 Why do businesses need an import license before customs clearance?
- 0.3 Common consequences when importing without a license
- 1 Latest legal basis in 2025 for medical equipment import license
- 2 Classification of medical equipment and conditions for applying for an import license
- 3 Procedures and documents for applying for a license to import medical equipment
- 4 Time, Fees and notes when applying for an import license
- 5 When permission is denied: Cause and treatment
- 6 Conclude
Overview of medical equipment import trends in 2025
2025, It is forecasted that the healthcare industry will continue to expand, especially in the field of importing medical equipment, due to the need to improve the quality of medical examination and treatment services and apply new technology. Imported medical equipment is increasingly diverse, from paraclinical equipment, diagnostic imaging to complex treatment support devices. That's why, compliance with legal regulations import medical equipment becomes extremely important to avoid legal risks as well as ensure sustainable business operations.

Why do businesses need an import license before customs clearance?
In activity import medical equipment, import license Acts as a mandatory legal certificate for state management agencies to allow businesses to bring goods into the market.. Specifically, import license help businesses:
- Meet the legal requirements of the Ministry of Health before importing medical equipment.
- Avoid being fined or having your goods detained at the border due to lack of valid licenses.
- Ensure products meet quality standards, safe according to regulations.
- Optimize customs clearance process, Minimize waiting time.
Common consequences when importing without a license
Intentional or unintentional importation medical equipment but not yet import license can lead to many serious consequences, include:
- Administratively sanctioned according to the provisions of law, can cause financial damage and corporate reputation.
- Goods are detained or destroyed according to regulations of the authorities.
- Delaying business plans, affecting distribution activities and customer service.
- Risk of criminal prosecution in case of serious consequences.
Understanding the application process import license help businesses avoid these risks, contributing to ensuring import activities take place smoothly, legal.
Fully following these instructions will help businesses clearly understand the process, Prepare documents fully and on time, thereby improving the likelihood of success in applying import license Medical equipment in 2025.
Latest legal basis in 2025 for medical equipment import license
In the context of the medical industry increasingly developing and innovating, updating related legal regulations License to import medical equipment is a key factor for businesses to operate legally and effectively. 2025, These regulations continue to be adjusted to comply with international trends and ensure safety, quality for users. Below are the main contents of the latest legal basis applicable to medical equipment import activities this year..

Law on medical equipment management: Decree 98/2021/ND-CP and Decree 07/2023/ND-CP
The current medical equipment management law is institutionalized through two important decrees:
- Decree 98/2021/ND-CP: Management regulations, classify, Quality check, safety and effectiveness of medical equipment, which clearly defines the conditions for import medical equipment conforms to national and international standards. This Decree has updated the regulations on classifying equipment according to group A, B, C, D, Expand technical standards, and import licensing process.
- Decree 07/2023/ND-CP: Continuation and revision, Additional content related to handling violations, inspection process, Certification and licensing procedures for importing medical equipment. This Decree also emphasizes the responsibility of businesses in complying with new regulations, Minimize legal risks in import activities.
These two decrees are the core legal foundation for operations import license medical equipment, clearly stipulate the conditions and procedures that businesses must comply with.
Current guiding circulars: Circular 05/2022/TT-BYT and latest update
In addition to the decrees, Related guidance documents also play an important role in guiding law enforcement:
- Circular 05/2022/TT-BYT: Is a document with specific instructions on the process, file, conditions to Apply for a license to import medical equipment. This Circular regulates the steps for submitting applications, check, appraisal and licensing, along with technical standards, The safety of medical equipment is in accordance with the latest regulations.
- The latest updates to this Circular include additions to documents proving origin, Consular legalization certificate, New technical standards for high-end medical equipment, as well as inspection regulations, post-licensing supervision.
These instructions help businesses understand the process and ensure compliance with legal regulations in the import of medical equipment., Avoid legal risks and handle violations.
Distinguish between import license and medical device circulation number
In the legal system about import medical equipment, It is necessary to clearly distinguish two important concepts:
- Import license: A certificate issued by a state management agency that allows businesses to bring medical equipment from abroad to Vietnam for circulation., distribute or use for a specific purpose. This is a mandatory condition to legalize import activities.
- Medical device circulation number: Is the certification number issued after the device is tested, technical assessment, Ensure compliance with safety standards, quality for distribution and use in the domestic market. The circulation number indicates that the device has been officially approved for circulation, The import license is only the first step in the distribution chain.
A clear distinction helps businesses avoid confusion during the permit application process, Ensure compliance with legal procedures and avoid legal risks related to import and distribution of medical equipment. Simultaneously, Understanding these regulations also helps businesses accurately identify documents, Documents need to be prepared to apply for an import license appropriate to each type of equipment.
So, 2025 continues to show clarity, Transparency in legal basis import license medical equipment, contributes to improving management efficiency, ensure community safety and promote medical equipment import activities in accordance with current legal regulations.
See more: Customs clearance services
Classification of medical equipment and conditions for applying for an import license
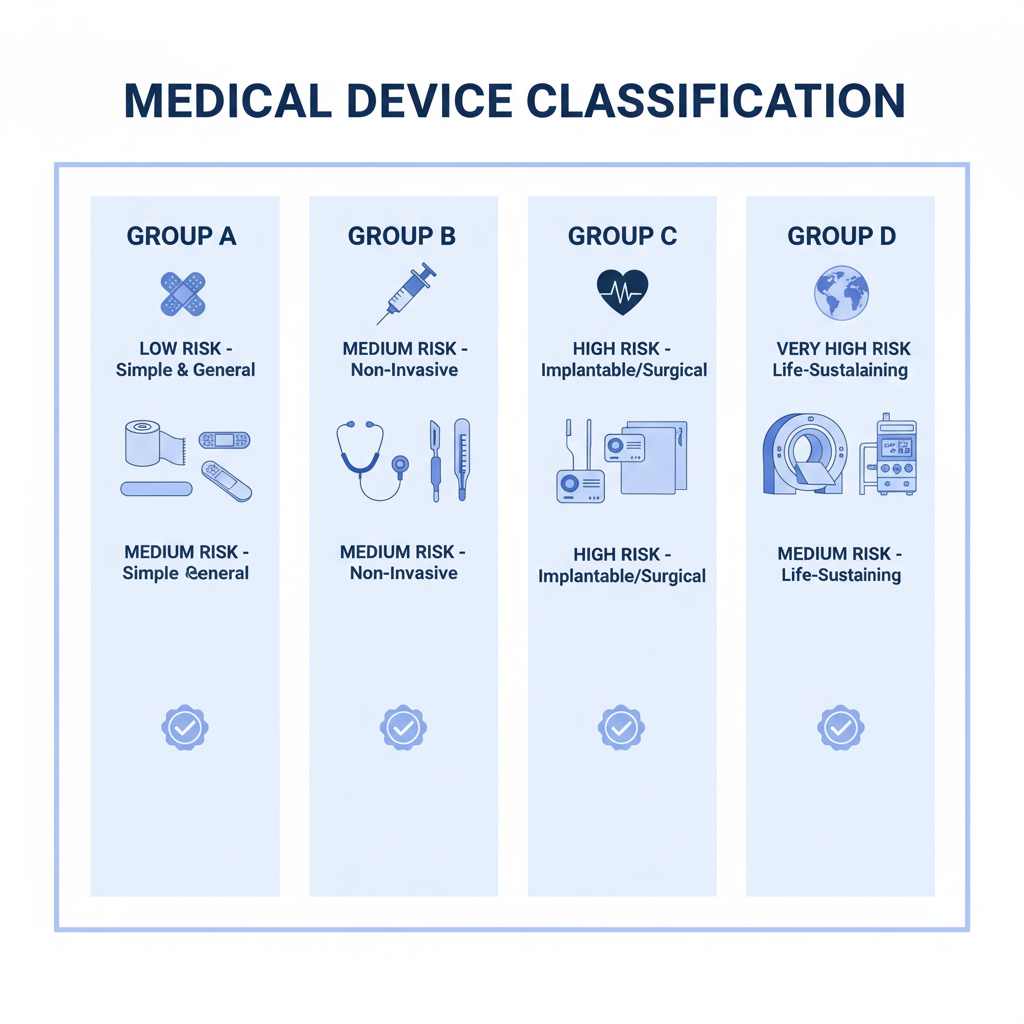
Identifying the correct medical device group is an important step in the application process import license medical equipment, to ensure compliance with legal regulations and avoid unwanted legal risks. In the management system of the Ministry of Health, Medical devices are classified into four main groups based on hazard level, nature of use and complexity of the device. How to determine the appropriate group helps businesses understand the necessary conditions to carry out import procedures.
How to identify medical device groups (Group A, B, C, D)
The determination of medical device groups is based on criteria of danger level, feature, and intended use. Specifically, The steps to determine are as follows:
- Investigate the properties of the device: Review the specification, main function, and device applications.
- Consider the level of danger: Assess the possibility of causing harm or affecting the user's health.
- Refer to the classification list of the Ministry of Health: Regularly update the latest regulations on equipment classification.
- Identify the right group: Based on analyzed criteria, Identify equipment belonging to group A, B, C or D.
These groups are classified as follows:
- Group A: Medical devices are less dangerous, Usually simple devices, Non-invasive or has little impact on the body.
- Group B: Average equipment, has the potential to cause impact but does not seriously affect the user's health.
- Group C: Equipment has a higher level of danger, often associated with invasiveness or major health impacts.
- Group D: The most dangerous device, often involves complex equipment, potentially dangerous if not used properly.
Group C devices & D is required to apply for an import license
In current regulations, Equipment in groups C and D are required import license before being put on the market or used in Vietnam. This requirement ensures these devices meet safety standards, quality and efficiency, as well as in accordance with strict import control regulations of the Ministry of Health.
These devices often include types such as::
- Complex image diagnostic equipment (MRI, CT scanner).
- High-end surgical equipment.
- Devices that support invasive or potentially dangerous treatment.
- Medical devices used in clinical trials or research.
This helps management agencies more closely control the quality of imported medical equipment, Limit risks related to safety and efficiency of use.
Illustrative examples for each group of medical devices
For easy visualization, Below are some illustrative examples for each group:
- Group A: Devices such as electronic thermometers, automatic blood pressure monitor, disposable medical equipment such as needles, bandage.
- Group B: Devices such as handheld ultrasound machines, rapid testing machine, Simple patient monitoring devices.
- Group C: Equipment such as digital X-ray machines, invasive mechanical ventilation equipment, endoscopic devices.
- Group D: Complex equipment such as MRI machines, CT machine, Robotic surgical system, devices that support highly precise medical intervention.
Identifying the correct equipment group helps businesses clearly define procedures, Documents need to be prepared and related legal conditions, thereby ensuring the import process goes smoothly, more legal and faster.
Procedures and documents for applying for a license to import medical equipment
The application import license Medical equipment is an important and mandatory step for businesses that want to carry out import activities in accordance with the law.. This process not only helps businesses ensure the legality of their goods but also avoids legal risks, fined or denied customs clearance at the border gate. Below are the specific steps in the process and the documents that need to be prepared to apply import license Medical equipment in accordance with the latest regulations in 2025.
Conditions of businesses and imported products (CO, CQ, CFS, Certificate of free circulation)
Before starting the application procedures import license, Enterprises need to meet the following conditions:
- Enterprises must have a license to operate business in the medical field, in accordance with the provisions of law.
- Imported products must meet technical standards, quality according to regulations, Have all documents proving legal origin such as:
- Certificate of origin (CO – Certificate of Origin)
- Quality certification (CQ – Certificate of Qualification)
- Certificate of free circulation (CFS – Certificate of Free Sale)
- These documents must be legal, valid and suitable for each specific type of medical device.
Documents need to be prepared: application form, authority, technical documents, valid documents
To carry out application procedures import license, Enterprises need to fully prepare the following types of documents:
- Application form for import license according to the form issued by the Ministry of Health.
- Power of attorney from businesses to individuals or organizations performing procedures.
- Technical documentation of medical devices: catalog included, Instructions for use, Certification of compliance with technical standards.
- Valid documents proving origin: CO, CQ, CFS, Certificate of free circulation.
- Other documents as required by competent authorities, as a certificate of international standards (ISO, CE, FDA) if any.
Note, Foreign documents need consular legalization in Vietnam according to regulations to ensure the validity of the documents.
Application process through the Ministry of Health Public Service Portal and steps to follow
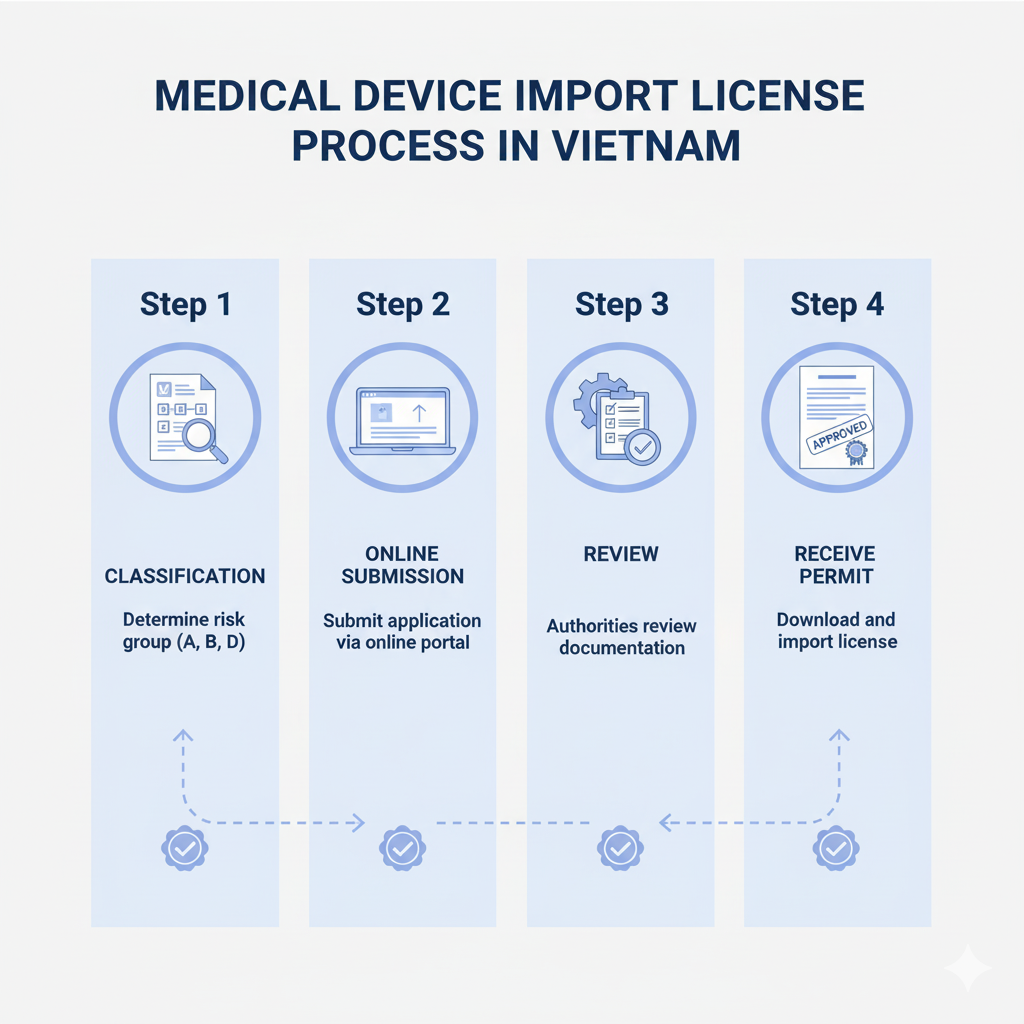
Application process import license Medical equipment through the Public Service Portal of the Ministry of Health includes the following main steps:
- Standardize records: Business check, review documents, document, Ensure completeness and validity according to the prescribed list.
- Login to the system: Access the Ministry of Health's Public Service Portal with your existing registered account.
- Fill in information and download profile: Fill in all necessary information and attach the prepared documents.
- Submit online application: Submit documents through the system and receive a confirmation code.
- Monitor processing progress: Update profile status, Respond or supplement documents if requested by the reviewing agency.
- Get results: When the application is approved, Businesses will receive notification of issuance import license through the system or via email.
- Print and archive: Print the license for use in import activities, Carry out customs procedures and store records according to regulations.
Correct implementation of this process helps businesses shorten processing time, Ensuring legal compliance and minimizing risks during the import of medical equipment.
Time, Fees and notes when applying for an import license
During the application procedure import license for medical equipment, understanding of time factors, fees as well as important notes will help businesses be more proactive in planning and minimize arising risks. Below are the contents to pay attention to so that the licensing process goes smoothly, effective.
Application processing time limit and how to shorten the approval time
According to current regulations, Processing time for import license application Medical equipment takes an average of 15 to 30 working days from the date of receipt of complete and valid documents. This time may be longer if the documents are missing or additional documents are needed, document. To shorten review time, businesses should:
- Prepare complete documents, legal and accurate from the start.
- Submit your application via Ministry of Health Public Service Portal to minimize administrative procedure time.
- Regularly monitor the system to update feedback from the licensing agency.
- In case of urgent need, Businesses can hire support services to speed up the processing process or request special consideration according to regulations.
Fees, service costs and how to optimize procedures
Related fees Apply for a license to import medical equipment usually included:
- Basic licensing fee according to regulations of the Ministry of Health.
- Service fee if the business hires consulting services or agents to carry out procedures.
- Fees related to inspection, quality certification (if required).
To optimize costs, businesses can:
- Prepare your own documents according to instructions from the Ministry of Health to reduce service costs.
- Refer to comprehensive services, There are clear time and cost commitments from reputable units.
- Choose the right support service to minimize errors, Avoid having to redo your application, thereby saving costs and time.
Common errors when applying for an import license and how to fix them
During the application process import license, Businesses often encounter some common errors such as::
- Wrong medical device group: causing delay or refusal of licensing. It is necessary to determine the correct device group based on the classification of the Ministry of Health.
- Documents are missing or invalid: Lack of documents such as authorization, Certificate of free circulation (CFS), certificate of origin (CO), quality certificate (CQ). To fix it, You need to carefully check the requirements and prepare fully before submitting your application.
- The dossier was not consularly legalized according to regulations: causing problems in document verification. Enterprises should properly follow the consular legalization process to ensure the validity of foreign documents.
- The information on the profile does not match: cause confusion or rejection. It is necessary to carefully check the data before submitting.
Paying attention to fixing these errors will help the licensing process go smoothly, Save time and cost.
Overall, understanding the factors time, fees and the Important note during the application process import license Medical equipment is a key factor for businesses to ensure legal compliance, Avoid late deadlines or refusal of permits. Practice correctly, Prepare complete documents, Choosing the appropriate support service will contribute to improving efficiency and minimizing risks during the import process.
When permission is denied: Cause and treatment
During the application process import license medical equipment, It is inevitable that a business's application will be rejected. Understanding the reasons leading to this decision and knowing how to handle it appropriately will help businesses quickly overcome remaining problems., avoid wasting time, costs and ensure compliance with legal regulations. Below is a detailed analysis of common causes as well as solutions when encountering this situation.
Common causes leading to refusal of import licenses
The refusal to grant a license often comes from the following main reasons:
- Missing or errors in records: Application form import license Incomplete or inaccurate information is the most common cause. This includes lack of necessary documents such as certificates of free circulation (CFS), certificate of origin (CO), quality certificate (CQ), or technical records are unclear.
- Does not fit medical device classification: Enterprises do not correctly identify medical equipment groups according to regulations, leading to documents that are not suitable or do not meet the specific requirements of each group.
- Medical equipment is not eligible for import: The equipment does not meet technical standards, do not have sufficient valid certification, or do not have a valid certificate of free circulation according to the law.
- Errors in legal documents or consular legalization: Documents from abroad are not valid, not consularly legalized according to the correct procedure, or lack of documents related to origin, quality.
- Failure to comply with the application process: Submitting documents through the public service portal incorrectly, Lack of checking steps or failure to update the latest records as required.
Instructions for revision, Supplement documents and reapply for license
When rejected, Businesses need to take the following steps to make amendments, Supplement documents and reapply import license effectively:
- Determine the exact cause of the refusal: Read carefully the decision from the licensing agency to clearly understand the reason. Normally, The agency will clearly notify you of any shortcomings or discrepancies in the records.
- Prepare additional documents, appropriate editing: Based on feedback content, Businesses need to update or supplement missing or invalid documents, as consular legalization certificate, Certification of compliance with technical standards, or documents related to product origin.
- Make necessary edits: Make sure all documents are correct, clear, full and valid according to the provisions of Decree 98/2021/ND-CP, Circular 05/2022/TT-BYT and latest instructions.
- Submit amended documents via the public service portal: After completing editing, Businesses need to submit applications via the Ministry of Health's electronic system, Make sure the steps are followed correctly.
- Contact directly to monitor progress: In case you need support or have questions, Businesses can directly contact the application processing officer to understand the process and receive quick feedback.
In case you need to re-apply for a license and steps to take
In some special cases, The application is refused or the license is revoked, Businesses are forced to reapply import license from the beginning. The steps are as follows:
- Clearly identify the reason for revocation or rejection: Read the management agency's announcement carefully to know the exact cause, From there, there will be appropriate treatment methods.
- Edit, Complete new profile: Secure all documents, certification, appropriate and valid documents as required, Additional new documents or certification of compliance with international standards may be required.
- Submit your application again: Follow the application process through the public service portal or directly at the competent authority.
- Wait for approval and monitor progress: During this process, Businesses need to regularly check the status of their documents, Ready to provide additional or clarify content if requested by the licensing agency.
- Get a new license After the application is successfully approved and full legal conditions are guaranteed.
Quick and correct handling of license refusal will help businesses save time, Avoid legal risks, At the same time, maintain legal and effective import of medical equipment.
[h2] Support service for applying for medical equipment import license from CUONG QUOC Logistics
[h3] Introducing comprehensive services, Professional working process of CONG QUOC Logistics
In an increasingly strict legal context and high requirements for the import of medical equipment, CUONG QUOC Logistics is proud to provide support services import license Professional medical equipment, package, Helps businesses save maximum time and effort in carrying out complex legal procedures.
We understand the importance of complying with legal regulations during the import of medical equipment, At the same time, we commit to providing services with clear processes, transparent, from the equipment classification step, Prepare documents for submission and track results. CUONG QUOC Logistics's working process includes main steps:
- Receive requests and analyze the type of medical equipment that needs to be imported
- Detailed consultation on conditions, documents need to be prepared
- Support businesses to prepare complete and valid documents according to regulations
- Submit application through the public service portal of the Ministry of Health
- Monitor, Update the review process and respond quickly to edit requests
- Receive import licenses and guide businesses on the next steps
We are proud to be a trusted partner of many businesses in the medical field, owns a team of experienced professionals, Always accurately update the latest regulations import license medical equipment.
[h3] Time commitment, costs and free consultation on procedures
CUONG QUOC Logistics is committed to providing customers:
- Fast processing time, ensure on schedule, Help businesses put medical equipment into operation soon
- Reasonable cost, clear, No hidden costs arise during service performance
- Free consultation, Support businesses to understand legal regulations, necessary conditions, Procedures need to be prepared for the application process import license went smoothly, smoothly
Besides, We also provide accompanying services such as checking the legality of documents, Instructions for editing rejected records, Helps businesses feel absolutely secure about the success of the application procedure import license medical equipment.
We understand that, Each customer has its own needs and characteristics, Therefore, CUONG QUOC Logistics's services are always flexible, Personalized according to each business's requirements. Our consulting team is always ready to listen, analysis, offer the most optimal solution, helps customers save time, costs and minimize legal risks in the process of importing medical equipment.
With the motto of taking customer satisfaction as the center, CUONG QUOC Logistics is committed to accompanying businesses throughout the application process import license, delivers quick results, exactly in accordance with current legal regulations in 2025.
Conclude
In the context of the healthcare industry increasingly developing and demanding high quality, Compliance with legal regulations related to importing medical equipment becomes a key factor to ensure business operations run smoothly and legally.. The article has detailed the trend of importing medical equipment in 2025, emphasized the importance of import licenses before customs clearance, as well as the serious consequences that can occur if the correct procedures are not followed.
The latest legal regulations such as Decree 98/2021/ND-CP, Decree 07/2023/ND-CP and guiding circulars such as Circular 05/2022/TT-BYT provide a solid legal foundation for businesses in determining equipment groups., Conditions for applying for permission and preparing complete documents.
Besides, The article also clearly guides the steps of the application process, processing time, Fees need to be prepared, along with common errors and how to handle problems when licensing is denied. Special, CUONG QUOC Logistics' support services help businesses save time, costs and minimize risks in the process of applying for a license to import medical equipment.

See more at